∞ generated and posted on 2016.08.25 ∞
Key waste product of the non-electron-transport aspects of cellular respiration.
Carbon dioxide is considered to be an oxidized, inorganic form of carbon. Sugars that enter glycolysis ultimately, in the course of cellular respiration, are converted to carbon dioxide and water, with their energy partially captured by ATP and their electrons captured by electron carriers such as NADH.
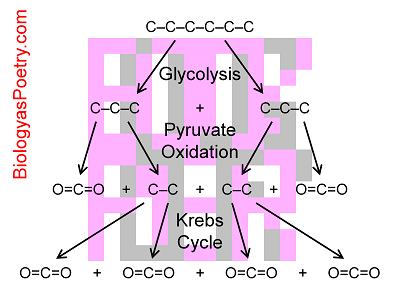
Figure legend: Shown is the splitting of a six-carbon sugar (hexose, usually glucose) into two three-carbon molecules (pyruvate) which are the converted into two-carbon molecules (acetyl CoA during pyruvate oxidation). This latter step generates two carbon dioxide (O=C=O times 2), one per pyruvate. Acetyl CoA is then fed into the Krebs cycle which generates four more carbon dioxide, two per acetyl CoA. In outline the reverse occurs in the course of photosynthesis. Numerous players in these reactions are not shown including hydrogen atoms, oxygen atoms, and acetyl CoA, all as attached to organic carbon atoms, e.g., C-C or C-C-C.
Carbon dioxide is a symmetrical and therefore nonpolar molecule despite its two carbon-to-oxygen bonds. In combination with its small size, it along with molecular oxygen (O2) are able to dissolve in lipid bilayers and therefore pass through membranes without protein facilitation.